The valence electrons will determine how many electrons are shared or transferred between atoms.When there is a large imbalance of electronegativity and ionization energy between atoms, an ionic bond will form.If we have a meta/nonmetal pairing then the nonmetal atom will pull strongly on the metal's weakly held (low ionization energy) electron and the metal atom will not pull very strongly (low electronegativity) on the strongly held (high ionization energy) electrons of the nonmetal atom.From the elements position on the periodic table, predict the valence shell electron configuration for each atom. Tins electron configuration is Kr5 s 2 4 d 10 5 p 2. When both atoms in the pair have a similar pull on the other's electrons (electronegativity) and resist the removal of an electron (ionization energy) in a similar way, then they will share electrons forming a covalent bond. Sn is located in the second column of the p block, so we expect that its electron configuration would end in p 2.In that situation the atoms will attract the other's electrons with about the same strength, and it will take a similar amount of energy to remove an electron from each atom. If we have a metal/metal or a nonmetal/nonmetal pairing then each atom in the pair will have a similar electronegativity and ionization energy as the atom it is bonded with. An atom with a closed shell of valence electrons (corresponding to an electron configuration (s2p6)) tends to be chemically inert.If two atoms are going to bond together we have three possible categories:. .svg/500px-Periodic_table_(polyatomic).svg.png)
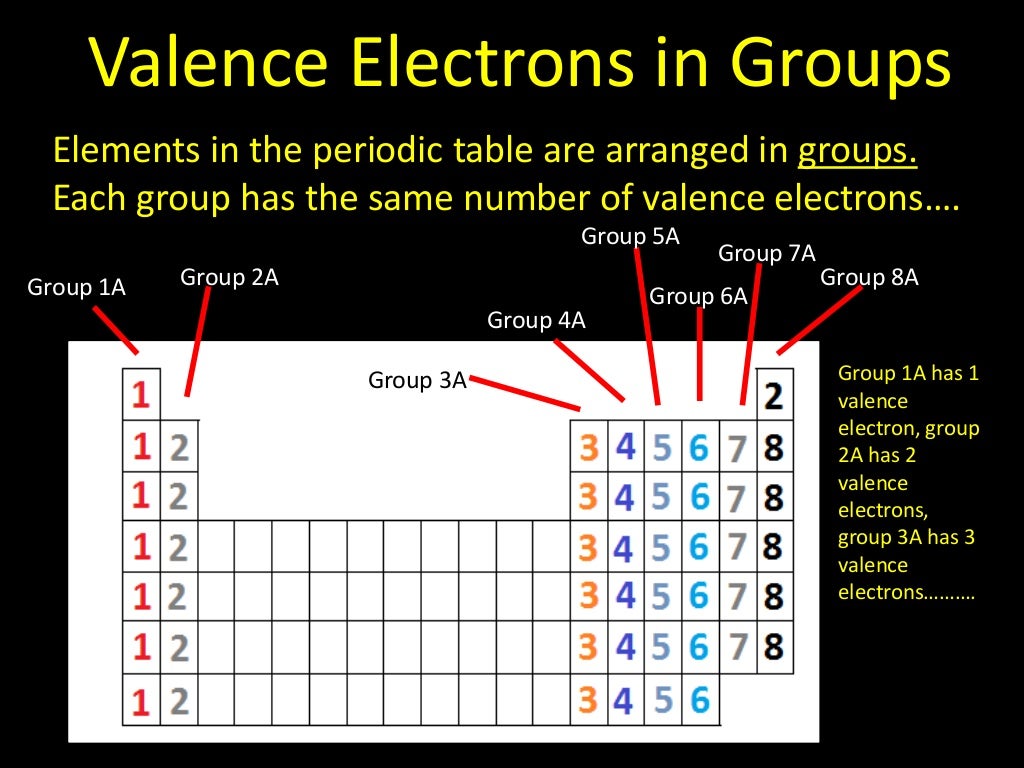
Non-Metals (high ionization energy and high electronegativity).Metals (low ionization energy and low electronegativity).The periodic table can be divided into two broad groups:.Using the variable n to represent the number of the valence electron shell, write the valence shell electron configuration for each group. Basically, atoms are most stable when they can achieve an outer electron structure similar to the closest Noble gas. The shape of the periodic table reflects the order in which electron shells and subshells fill with electrons.This can be done by either sharing electrons between atoms, taking electrons from other atoms, or giving electrons away to other atoms. If atoms don't have 8 valence electrons (or two if they are close to He), then they will react with other atoms in order to have 8 valence electrons.One exception is He which has a full principle energy level with 2 electrons. These atoms are often called the alkali and alkaline earth elements. For example, potassium has a configuration Ar4s 1. 1 ), the configuration is a closed shell of core electrons, plus s electrons in a new shell. All of these elements have 8 valence electrons in their highest principle energy level. For atoms found in the first two columns of the periodic table (figure 1.6.1 1.6. The electron configurations of the elements are in Figure 6.9.2. One simple piece of evidence for this is the Noble Gases which form the last column on the right of the periodic table. 1: The Periodic Table, Showing How the Elements Are Grouped According to the Kind of Subshell ( s, p, d, f) Being Filled with Electrons in the Valence Shell of Each Element.Experimental evidence shows us that atoms are most stable when they have full s and p orbitals (8 valence electrons) in their highest principle energy level.Pick two elements from any column and determine how many valence electrons those atoms have.Bromine has 7 valence electrons: 1s 22s 22p 63s 23p 6 4s 23d 10 4p 5.Sodium has 1 valence electron: 1s 22s 22p 6 3s 1.Oxygen has 6 valence electrons: 1s 2 2s 22p 4.It is these electrons that primarily interact with other atoms. 1: Periodic table by Dmitri Mendeleev, 1871. After the development of quantum mechanics, it was shown that the halogens all had seven valence electrons, supporting their original placement into the same group on Mendeleev's periodic table. Valence electrons are those electrons that are in the highest principle energy level. As a gas or vapor, the halogens all had a pungent odor.Within a group (family) of elements, atoms form ions of a certain charge. The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
